In the post-epidemic era of the blue ocean market--POC, Screen, Home Testing,Nanjing Synthgene Medical accelerates product line distribution!
Release time:
2021-10-24
Since the beginning of this year, the global COVID-19 epidemic has not slowed down due to the rapid vaccination of the vaccine. The global anti-epidemic situation is still severe. Different countries are experiencing the second, third or fourth peak. Statistics show that Until October 24, 2021, the cumulative number of reported cases of new COVID-19 infection worldwide has exceeded 240 million. The new COVID-19 anti-epidemic will be a protracted battle in the world!
Since the beginning of this year, the global COVID-19 epidemic has not slowed down due to the rapid vaccination of the vaccine. The global anti-epidemic situation is still severe. Different countries are experiencing the second, third or fourth peak. Statistics show that Until October 24, 2021, the cumulative number of reported cases of new COVID-19 infection worldwide has exceeded 240 million. The new COVID-19 anti-epidemic will be a protracted battle in the world!
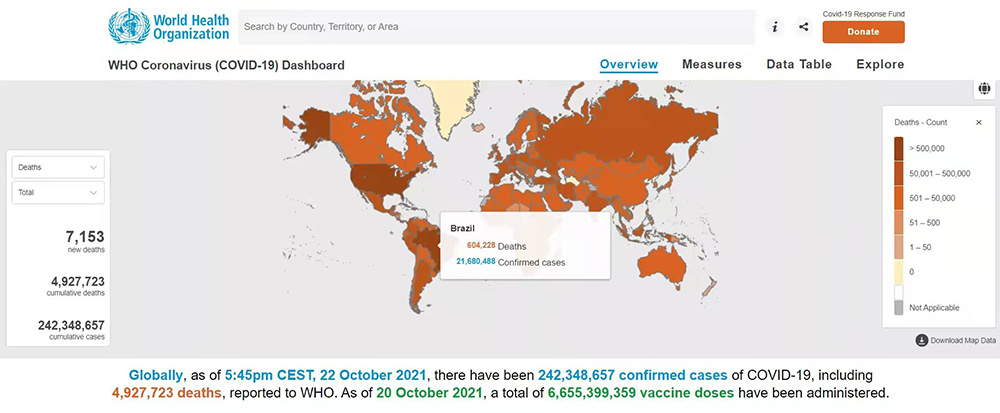
Containing the spread of the epidemic is inseparable from rapid and effective virus detection methods. The main reason why South Korea was successful in fighting the spread of the epidemic in the early days was the large number of tests. In contrast, although the United States has many high-tech companies, it also developed a large number of test kids in the early stages of the epidemic. but the early government did not pay much attention to testing. When the first wave of the epidemic spread, the best time to control the epidemic was missed. After Biden came on stage, the United States began to pay attention to the importance of testing. FDA has recently begun to focus on encouragement and approval of New detection reagents for POC, Screening and Home testing.
The sudden COVID-19 epidemic has sounded the alarm for us, and quick and easy testing will also become a normalized response. This is true for the COVID-19, as are other infectious diseases.
Quick and easy screening tests can be popularized in communities, offices, schools for asymptomatic testing, and can also be placed in terminal medical institutions (Point of Care), or at home without a doctor’s prescription (OTC) for quick and effective test.
Two quick and easy test reagents,
According to the FDA, there are two types of quick and easy test reagents:
The first is a quick disposable colloidal gold reagent, similar to a pregnancy test stick, suitable for testing at home, and the results are usually available in about 15 minutes.
The second type is a portable and easy-to-operate fluorescence analyzer. This type of tester can be carried and is cheap and does not require professional operation. The results can usually be obtained in 15-30 minutes. It can be popularized in communities, enterprises, schools, and can also be placed in terminal medical institutions (Point of Care). POC testing and home testing will be the development direction of the reagent market. According to a research report by Research And Markets, the global POC market will reach 45 billion US dollars by 2025, which will be nearly double the 29 billion US dollars in 2020. In October last year, Cue Health, a US biotech company based in San Diego, received a large purchase order from the US government of $480 million. Cue Health was the only company approved by the FDA EUA to produce portable POC nucleic acid detectors. According to a report from the US financial channel CNBC, the price of the nucleic acid detector that Cue Health can carry is several hundred dollars, and the price of each test kit is tens of dollars.
Therefore, the development of detection reagents for POC, Screening and Home testing is a direction with huge market potential. Nanjing Synthgene Medical is currently accelerating its product line layout and get ready to enter this blue ocean market!
1: colloidal gold testing products suitable for screening asymptomatic people in communities, offices, and schools
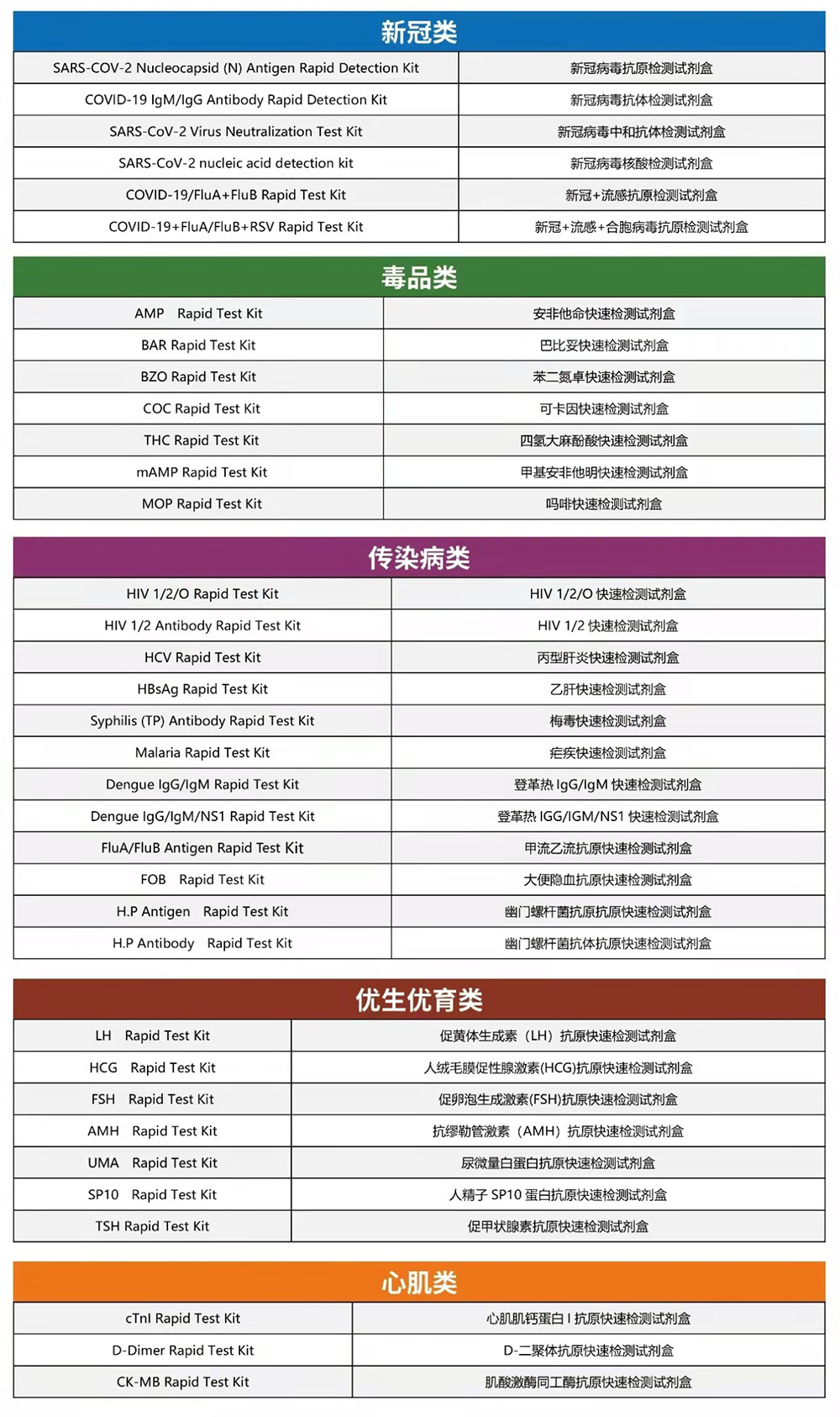
2: Quantum dot fluorescence tomography products suitable for testing in terminal medical institutions

3、intelligent testing products suitable for home self-testing



