Neisseria Gonorrhoeae Antigen Rapid Test Kit

This product is used for in vitro qualitative detection of Neisseria gonorrhoeae (NGH) in female cervical swabs and male urethral swabs. It is used for auxiliary diagnosis of Neisseria gonorrhea infection and is only used for in vitro diagnosis.
TEST TYPE: Female cervical swabs and male urethral swabs
RESULT IN: 15-30 minutes
Ordering Information
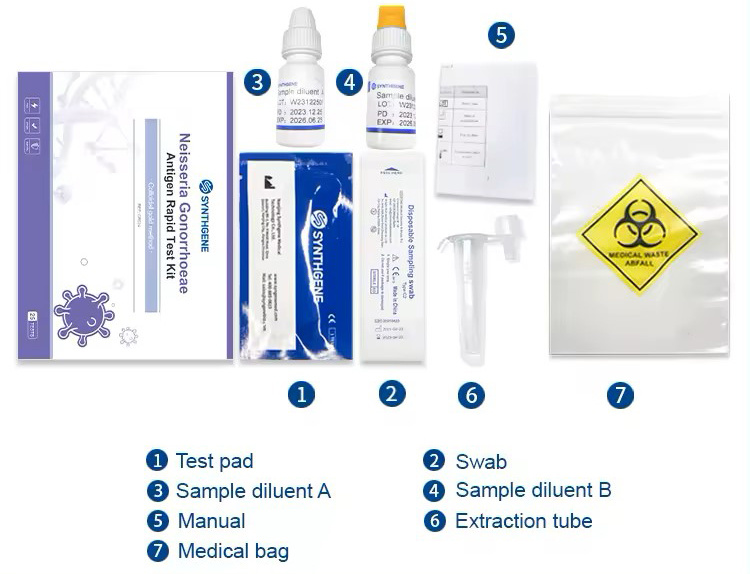
Specifications and performance
|
TEST TYPE |
female cervical swabs and male urethral swabs. |
|
CUT-OFF (DETECTION LEVEL) |
The enterprise limit of detection reference products (S1-S3) are tested, S1 is positive, S2 can be positive or negative, and S3 is negative. |
|
RESULTS IN |
15-30 minutes |
|
RELIABILITY |
Excellent (≈99%) |
|
MANUFACTURER |
SYNTHGENE |
About this test
Gonorrhea is a purulent infection of the genitourinary system caused by Neisseria
gonorrhoeae. It is one of the most common venereal diseases, and its main manifestations are gonococcal urethritis and cervicitis. Neisseria gonorrhoeae can also spread infection locally through the urethra or cervix, causing epididymitis and pelvic inflammatory disease; it can also cause disseminated gonorrhea through hematogenous transmission. In addition, it can also cause infections of the eyes, pharynx, and rectum; although some patients have been infected by gonorrhea, they have no clinical symptoms and become asymptomatic gonorrhea.
Commonly used laboratory methods for the diagnosis of gonococcal infection are as
follows: Specimen smear staining and direct microscopic examination; Cell culture
method; Antigen detection test, including direct immunofluorescence method and
enzyme-linked immunosorbent assay; DNA hybridization and polymerase Chain reaction (PCR) method; Serum antibody detection.
Quick and easy to use
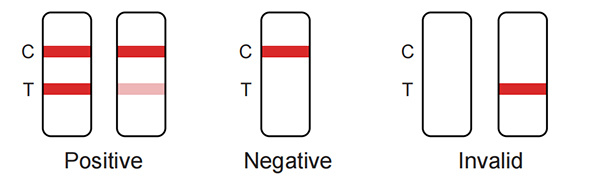
Positive: Red bands appear in both the test area(T) and the quality control area (C). The results show that the samples contain Neisseria gonorrhoeae antigen.
Negative: There is no red band appears in the test area (T), and a red band appears in the quality control area (C). The results show that Neisseria gonorrhoeae antigen is not detected in the samples.
Invalid: There is no red band in the quality control area (C), regardless of whether there is a red band in the test area (T), indicating the test result of the kit is invalid, and it is recommended to retest.
Product inquiry
We will contact you within one working day. Please pay attention to your email.
Related Products


