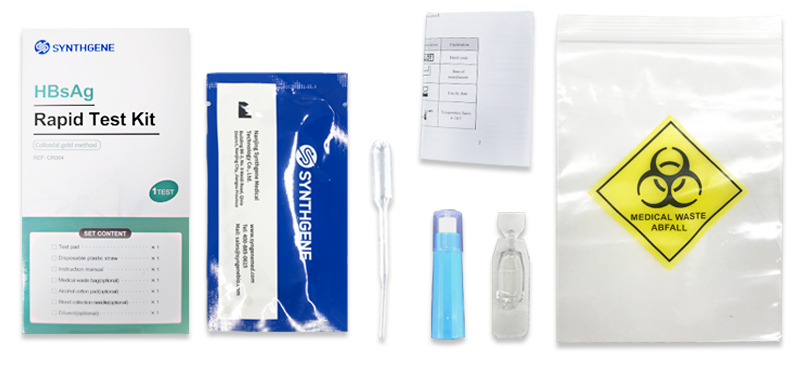
HBsAg Rapid Test Kit

Hepatitis B virus surface antigen detection can detect hepatitis B virus surface antigen in human whole blood, serumor plasma samples within10-30 minutes.
It is suitable for auxiliary diagnosis of hepatitis B virus infection.
Test Type:Whole blood (fingertip blood/venous blood), serum, plasma.
Detection time: 10-30 minutes
Ordering Information
Specifications and performance
|
Test Type: |
whole blood (venous or fingertip blood), serum or plasma |
|
Detection time: |
10-30 minutes |
|
Limit of Detection (LOD): |
1 IU/ml |
|
Reliability: |
Excellent |
|
Manufacturer: |
Nanjing Synthgene Medical Technology Co., Ltd. |
Operation process
About this test
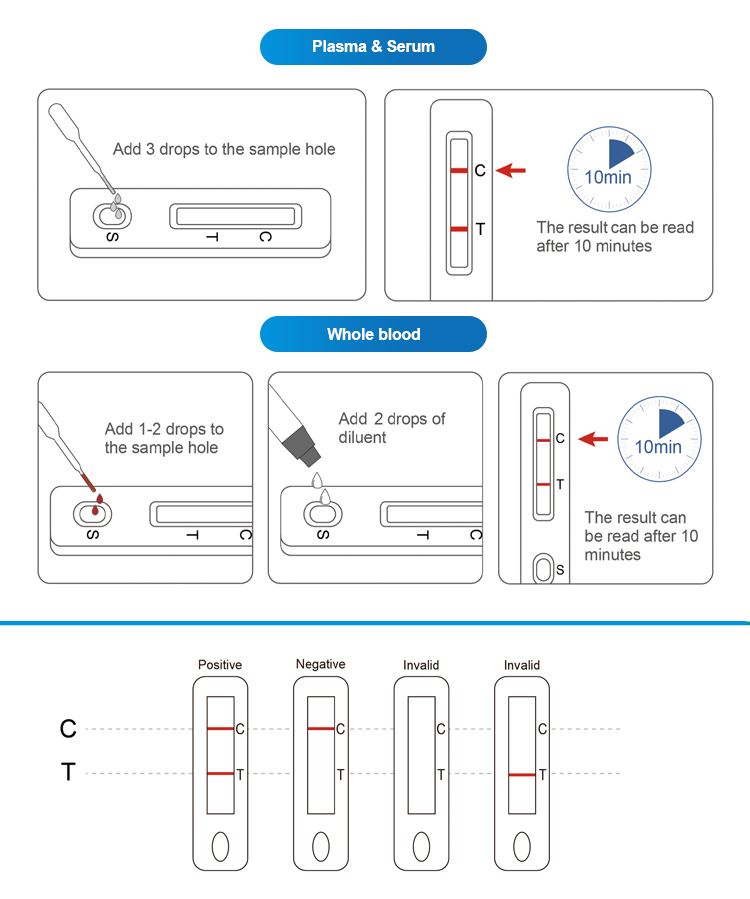
This product is suitable for the auxiliary diagnosis of hepatitis B virus infection. Hepatitis B is a worldwide infectious disease caused by hepatitis B virus (HBV), which is mainly transmitted through blood, mother-to-child and sexual contact. Hepatitis B surface antigen is the coat protein of hepatitis B virus, which appears in the blood with hepatitis B virus infection and is the main sign of hepatitis B virus infection. Hepatitis B surface antigen detection is one of the main detection methods for this disease.The product only requires three drops of human plasma/spine (approximately 75ul) and two drop of human whole blood (fingertip blood or venous blood) (approximately 50ul). The results will be observed in 10 minutes and will be effective within 30 minutes.
Fast and easy to use
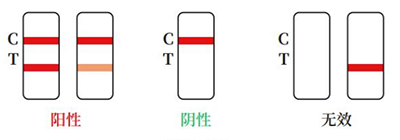
Positive:Red bands appear in both the test area (T) and the control area (C). The results show that the sample contains HBsAg.
Negative:There is no red band in the test area (T), and a red band appears in the control area (C). HBsAg is not detected in the sample
Invalid:No red band appears in the control area (C), regardless of whether there is a red band in the test area (T), the test pad is judged to be invalid, and it is recommended to perfo-rm a retest.
Operation Suggestions
1. This product is a disposable in vitro diagnostic product, please use it within the validity period.
2. Please read the instruction manual carefully before use, the inspection personnel must be professionally trained to operate, and the test operation should be carried out in strict accordance with the instructions of the kit.
3. If you find that the aluminum foil bag packaging is damaged, please do not use it Please use it as soon as possible after opening the aluminum foil bag. Do not conduct experiments in an excessively high temperature or humidity and excessive ly dry environment. The low temperature storage kit needs to be restored to room temperature before opening to avoid moisture absorption.
4. The test of the sample must be carried out in a specific environment, and the blood sample contacted during the test should be operated in accordance with the inspection procedures of the infectious disease laboratory.
5. Acute or chronic HBV infection should be reported to the local Center for Disease Control (CDC) in a timely manner in accordance with the Law of the local regulations on Prevention and Control of Infectious Diseases, and should indicate whether it is acute hepatitis B or chronic hepatitis B.
6. The test sample should avoid repeated freezing and thawing, and the sample with bacterial contamination should not be used for the test, so as not to affect the test result. Samples stored at low temperatures need to be equilibrated to room tempera-ture before use.
7. It should be kept clean, and pollutants should be treated as waste. The waste treatment is carried out in accordance with the local regulations for the safe disposal of waste and the infectious waste. Please handle with care.
Operation Flow
Product inquiry
We will contact you within one working day. Please pay attention to your email.
Related Products